We should also mention the Honda Clarity and the Hyundai Tucson FCEV, all three of which are very expensive cars - at around 60,000 euros - that are produced in drip-feed production and are hardly in the hands of private individuals. These problems, coupled with a dedicated refuelling infrastructure that is almost non-existent outside of a few specific locations, don't exactly make them attractive to the general public.
Let's take a look at how it works to understand it better.
What is a fuel cell
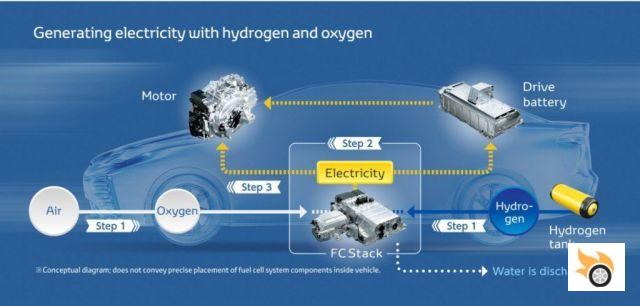
Basically it is a device that, thanks to a fuel and an oxidant, generates a chemical reaction of oxidation at the anode that releases electrons, which pass through the electrolyte, recombine at the cathode through reduction and, therefore, produces electricity. To make it easy, we can say that two chemical elements enter, and as a result we get electricity and another chemical element. In the case of vehicles, Hydrogen (H2) and air (O2) go in, which reacts through an electrolyte to deliver electricity and water (H2O).
Fuel cell vehicles are electric, only that instead of accumulating all the energy obtained from an external source (plug or solar panel) they generate it on board with a fuel that will need to be refueled. They have an obvious advantage in autonomy, because they make more kilometers per "recharge" and that takes a few minutes. On the other hand, they have the obvious disadvantage that they need a very high purity hydrogen, otherwise the delicate and expensive materials that make up the batteries are contaminated.
To understand it better, I leave you a very explanatory video about hydrogen fuel cells:
Types of fuel cells
Depending on the fuel used, they can be hydrogen, methanol, ethanol or glucose. In the case of vehicles, the most common fuel is hydrogen, but the most common classification is according to the electrolyte used.
- Fuel cells with PEM membranes (Proton Exchange Membrane): These are the most commonly used in the automotive industry (DuPont usually manufactures the membrane), mainly because they operate at low temperatures, i.e., between 60 and 80 ºC. The moving charges are H+ ions.
- AFC (Alkaline Fuel Cells): In this case they work at a higher temperature, around 120ºC. In this case the charges that pass through the catalyst are OH- ions.
- PAFC fuel cells (Phosphoric Acid Fuel Cells): They work at high temperature, around 220 ºC, they use phosphoric acid as electrolyte, and the charges in movement are H+.
- MCFC fuel cells (Molten Carbonate Fuel Cells): These are used in large power generation plants. They operate at very high temperatures, between 600 and 700 ºC, and the charges in motion are CO32-.
- SOFC (Solid Oxide Fuel Cells): Very similar to the previous ones, they operate between 600 and 1,000 ºC, and are used in large installations as heat recuperators. The moving charges are O2- ions. Nissan introduced this year a prototype e-NV200 powered by a bioethanol SOFC cell.
The use of hydrogen guarantees the cleanest emissions, only water vapour, the other solutions generate more waste.
The application in vehicles
And how do we apply this to a vehicle? Well, the truth is that a lot has changed since GM launched its Electrovan. The components have become so simplified and miniaturised that their packaging now fits into almost any platform. In general, a hydrogen fuel cell vehicle is basically an electric vehicle, many components are common to battery-powered models. In general, it consists of the following parts:
- Fuel tanks (hydrogen)
- Hydrogen fuel cell
- Backup batteries
- One or more electric motors
The fuel passes through the air, through the hydrogen fuel cell, and generates electricity and water. This charges the backup battery and drives the vehicle through the electric motors.
As you will understand, it is the hydrogen tanks that take up the most space, as they are pressurised cylinders, which must keep the gas in a liquid state to minimise the volume they occupy. Throughout the evolution of the hydrogen fuel cell car, they have been the great headache of the designers. Compressed hydrogen has a very low density per unit mass (it weighs very little) but takes up a lot of space because of its low density per unit volume.
Refilling its tanks takes no more time than filling up with petrol. Of course, although it already has very high safety levels, handling hydrogen is still dangerous... and weird. Has anyone seen a hydrogen plant? And this brings us to the next question: why hasn't this technology succeeded?
The future
Despite having invested more than 2,500 million dollars, 50 years of effort and having done more than 5 million kilometers in real conditions in the case of General Motors, it has not taken off. And there are two fundamental factors that prevent it from taking off: the development of the refuelling infrastructure is very scarce and the price of the components, and therefore of the vehicles, is very high.
If we add to this that the electric car is maturing very quickly, and lowering its price, I get the feeling that hydrogen cars are the equivalent of the Betamax videos of the 70-80s: they have failed before being born ... but not completely. It seems that there are niches where they can be very interesting, such as military applications (see GM's ZH2) or heavy transport, both for passengers (the Toyota H2, created for the 2020 Tokyo Olympics) and goods (Nikola One, Renault Maxity).
In any case, whether it succeeds or not, we should be grateful for the R&D efforts made by some companies such as GM, to take us to the next generation of non-polluting vehicles.
Manufacturers believe that it will be a technology that will coexist with pure electric vehicles in a future in which fossil fuels have lost their prominence, i.e. beyond 2030. The fuel cell makes more sense in larger vehicles or those that are going to travel longer distances, while pure electric vehicles are more suitable for short or medium distances. They are therefore complementary technologies, one does not replace the other.